Molecules whose solutions can carry electric current. ionize in water. Which of the following does not increase the rate at which a solid dissolves in water. using large pieces of the solid. Henry’s law relates. pressure to gas-liquid solubility. Raising the temperature of a solvent causes solvent-solvent coltisions to become.
BRAVE NEW WORLD Ch. 7-12 Quote Map (Quiz, Worksheet, Review, Study) – NO PREP
In Chapter 12 of the modern chemistry textbook, students are introduced to the concept of solutions. This chapter explores various aspects of solutions, such as types of solutions, concentration, and colligative properties. In order to assess their understanding of this topic, students often have to complete review questions.

Source Image: stuvia.com
Download Image
Study with Quizlet and memorize flashcards containing terms like According to Newton’s 2nd Law of Motion, Acceleration = Net Force divided by _____, According to Newton’s 3rd Law, for EVERY action, there is an equal and opposite _____, The product of an object’s mass and velocity is its _____ and more.
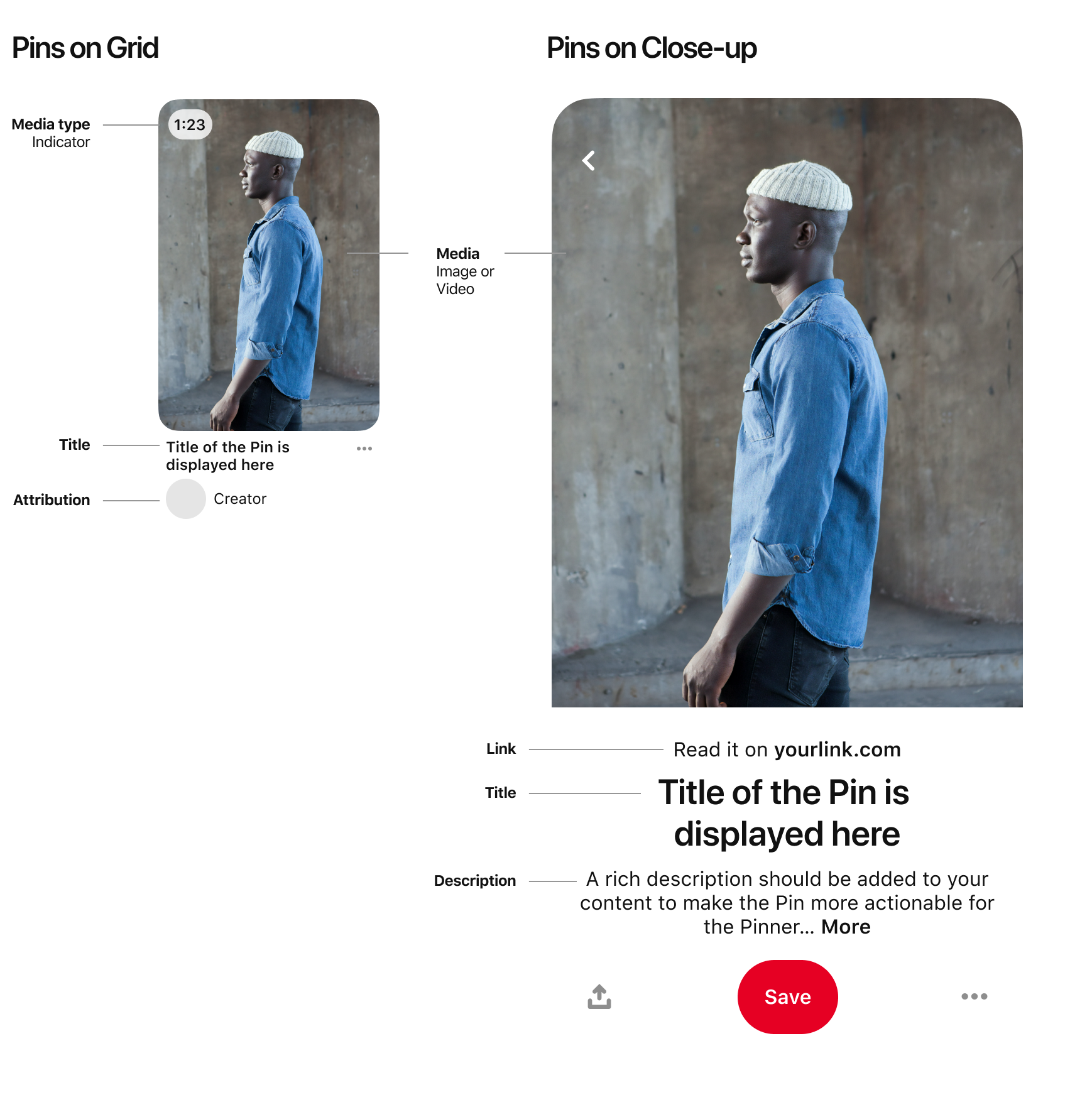
Source Image: help.pinterest.com
Download Image
A Year of Growth and Innovation: LearnWorlds 2023 Year in Review | LearnWorlds Blog Student Text, p. 580, Chapter 12 Review Answers: Student Text Page: Adobe Acrobat (.pdf) Teacher’s Resources, p. 337: Teaching Notes: Adobe Acrobat (.pdf) Assessment Rubric 1: Knowledge/ Understanding: Assessment Tool: Adobe Acrobat to word-processed: Assessment Rubric 2: Inquiry: Assessment Tool: Adobe Acrobat to word-processed: Assessment

Source Image: ptpioneer.com
Download Image
Chapter 12 Review Solutions
Student Text, p. 580, Chapter 12 Review Answers: Student Text Page: Adobe Acrobat (.pdf) Teacher’s Resources, p. 337: Teaching Notes: Adobe Acrobat (.pdf) Assessment Rubric 1: Knowledge/ Understanding: Assessment Tool: Adobe Acrobat to word-processed: Assessment Rubric 2: Inquiry: Assessment Tool: Adobe Acrobat to word-processed: Assessment 1. The following are statements about the dissolving process. Explain each one at the molecular level. a. Increasing the pressure of a solute gas above a liquid solution increases the solubility of the gas in the liquid. b. Increasing the temperature of water speeds up the rate at which many solids dissolve in this solvent. c. Increasing the
ISSA Nutritionist Chapter 12: Client Assessment and Goal Setting
Modern Chemistry 107 Solutions CHAPTER 12 REVIEW Solutions SECTION 3 SHORT ANSWER Answer the following questions in the space provided. 1. Describe the errors made by the following students in making molar solutions. a. James needs a 0.600 M solution of KCl. He measures out 0.600 g of KCl and adds 1 L of water to the solid. Intro to chem Chapter 12 solutions – YouTube

Source Image: m.youtube.com
Download Image
Essential Guide for Mental Performance Consultants (Digital Resource), The – Human Kinetics Modern Chemistry 107 Solutions CHAPTER 12 REVIEW Solutions SECTION 3 SHORT ANSWER Answer the following questions in the space provided. 1. Describe the errors made by the following students in making molar solutions. a. James needs a 0.600 M solution of KCl. He measures out 0.600 g of KCl and adds 1 L of water to the solid.

Source Image: us.humankinetics.com
Download Image
BRAVE NEW WORLD Ch. 7-12 Quote Map (Quiz, Worksheet, Review, Study) – NO PREP Molecules whose solutions can carry electric current. ionize in water. Which of the following does not increase the rate at which a solid dissolves in water. using large pieces of the solid. Henry’s law relates. pressure to gas-liquid solubility. Raising the temperature of a solvent causes solvent-solvent coltisions to become.

Source Image: teacherspayteachers.com
Download Image
A Year of Growth and Innovation: LearnWorlds 2023 Year in Review | LearnWorlds Blog Study with Quizlet and memorize flashcards containing terms like According to Newton’s 2nd Law of Motion, Acceleration = Net Force divided by _____, According to Newton’s 3rd Law, for EVERY action, there is an equal and opposite _____, The product of an object’s mass and velocity is its _____ and more.

Source Image: learnworlds.com
Download Image
Pinterest Modern Chemistry 103 Solutions CHAPTER 12 REVIEW Solutions SECTION 1 SHORT ANSWER Answer the following questions in the space provided. 1. Match the type of mixture on the left to its representative particle diameter on the right. _____ solutions (a) larger than 1000 nm _____ suspensions (b) 1 nm to 1000 nm

Source Image: facebook.com
Download Image
White Chocolate Bark (5 minute prep) – Two Kooks In The Kitchen Student Text, p. 580, Chapter 12 Review Answers: Student Text Page: Adobe Acrobat (.pdf) Teacher’s Resources, p. 337: Teaching Notes: Adobe Acrobat (.pdf) Assessment Rubric 1: Knowledge/ Understanding: Assessment Tool: Adobe Acrobat to word-processed: Assessment Rubric 2: Inquiry: Assessment Tool: Adobe Acrobat to word-processed: Assessment

Source Image: twokooksinthekitchen.com
Download Image
Chapter 12 Preview Objectives – ppt download 1. The following are statements about the dissolving process. Explain each one at the molecular level. a. Increasing the pressure of a solute gas above a liquid solution increases the solubility of the gas in the liquid. b. Increasing the temperature of water speeds up the rate at which many solids dissolve in this solvent. c. Increasing the

Source Image: slideplayer.com
Download Image
Essential Guide for Mental Performance Consultants (Digital Resource), The – Human Kinetics
Chapter 12 Preview Objectives – ppt download In Chapter 12 of the modern chemistry textbook, students are introduced to the concept of solutions. This chapter explores various aspects of solutions, such as types of solutions, concentration, and colligative properties. In order to assess their understanding of this topic, students often have to complete review questions.
A Year of Growth and Innovation: LearnWorlds 2023 Year in Review | LearnWorlds Blog White Chocolate Bark (5 minute prep) – Two Kooks In The Kitchen Modern Chemistry 103 Solutions CHAPTER 12 REVIEW Solutions SECTION 1 SHORT ANSWER Answer the following questions in the space provided. 1. Match the type of mixture on the left to its representative particle diameter on the right. _____ solutions (a) larger than 1000 nm _____ suspensions (b) 1 nm to 1000 nm
